Antimicrobial Additives Agent – Inorganic
1.Product Introduction
GERM ARMOR® antimicrobial materials can also continuously catalyze the conversion of water and oxygen to reactive oxygen species (ROS) to kill microbe via non-contact killing mechanism. It could generate different type of ROS, targeting different applications.
GERM ARMOR® antimicrobial materials have no drug resistance development, no chemical releasing, and no environmental contamination. It has high efficacy against various bacteria (Superbugs), fungi and virus.
GERM ARMOR® antimicrobial materials also have long-term activity, its lifetime could be as long as 10 years.
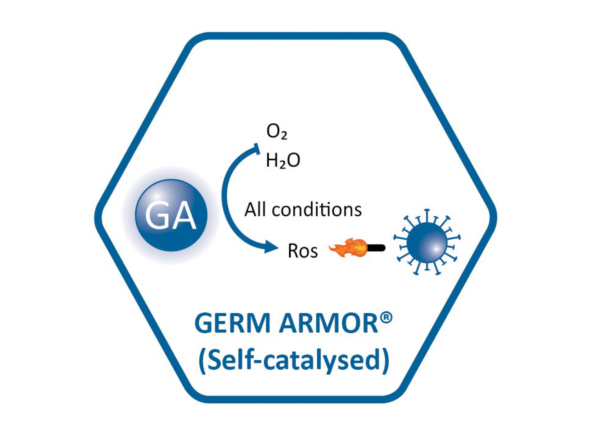
2.Product Parameter
Table 1 General properties
Items | Value |
Density | 2.1~3.5 g/cm3 |
Melting point | >400 ℃ |
Boiling point | >900 ℃ |
PH | 7-8 |
Particle Size | Adjustable |
3.Product Feature and Application
● Coatings and Paints
● Plastics
● Fibres
● Textiles
● Sanitizing water
● Others (Food application/Agriculture)

4.Products lists

| Applications | Effect |
| Product | Type | Particel size | Appearance | Water solubility | S.G (g/cm3) | Paints | Coatings | Plastics | Textiles | Rubber | Direct loading/Contact | Sanitizing | Bacteria | Virus | Fungi |
| GA 01 | Zinc-based | | Grey powder | No |
| | | | | |
| | |
| |
| GA 02 | Iron-based | | Black powder | No |
|
|
|
|
|
| | | | | |
| GA 03 | Zinc-based-Enhanced | | Grey powder | No | 2.5 | | | | | |
| | | | |
| GA 04 | Iron-based-Enhanced | | Black powder | No |
|
|
|
|
|
| | | | | |
| GA 05 | Copper-based | | Brown powder | No |
| | |
|
|
|
| | | | |
| GA Coating-1 | WB PAA based | N/A |
| N/A |
|
|
|
|
|
|
|
| | | |
| GA Coating-2 | WB PU-based | N/A |
| N/A |
|
|
|
|
|
|
|
| | | |
| Applications | Effect |
| Product | Type | Particel size | Appearance | Water solubility | S.G (g/cm3) | Paints | Coatings | Plastics | Textiles | Rubber | Direct loading/Contact | Sanitizing | Bacteria | Virus | Fungi |
| GAP 1 | Polyquats | N/A | White powder | Yes | N/A |
|
| | | |
|
| |
| |
| GAP 2 | Polyimidazolium | N/A | White powder | Yes | N/A |
|
| | | |
|
| | | |
| GAP 3 | Polyquats-modified | N/A | White powder | Yes | N/A | | |
|
|
|
|
| |
| |
| GAP 4 | Polyimidazolium-modified | N/A | White powder | Yes | N/A | | |
|
|
|
|
| | | |
| GAP 01 | Water/alcohol based solution | N/A | Transparent | N/A | N/A |
|
|
|
|
| ● |
| | | |
| GAS 02 | Water based solution | N/A | Transparent | N/A | N/A |
|
|
|
|
| ● |
| |
| |
5.Product Qualification
1. Passed Acute Toxicity Test
Testing method: mice oral/skin administration
Medium Lethal Dose (LD50):
for oral administration: LD50 > 2000 mg/kg.
for skin topically administration: LD50 > 10000 mg/kg.
2. Passed Geno Toxicity Test
Testing method: Genotoxicity and mutagenicity bacterial reversion assay (Ames)
Tested by: PWG genetics Pte Ltd
Conclusion: Non-mutagenic
3. Passed Skin Sensitivity Test
Testing method: Buehler test on guinea pigs (patch)
Tested by: TUV SUD (GLP lab)
Conclusion: GERM ARMOR® antimicrobial materials did not produce skin sensitization (level 0)




